- Blog
- Rab ne bana di jodi 720p google drive
- Mighty gunvolt burst icons
- Tekkera cinematic strings 2
- Ekla chalo re song meaning in hindi
- How to crack accounts with sentry mba
- Buy rapelay
- Trials fusion for ps3
- Creating visual studio extensions
- Snap on modis generic
- Mojave darwin-iso
- Bcs class 1 drugs
- Infinity sign tg ehentia
- Sonarr setup tutorial
- The owl house season 1 episode 6

This model can assess passive permeability, a current prerequisite for nonclinical classification. Submission of the data from a BCS biowaiver to the FDA, as well as the ANDA review and approval process, is similar to those with data obtained by regular in vivo experiments.Ībsorption Systems’ in vitro test systemsĪt Absorption Systems, we have employed a patented in vitro test system based on the Caco-2 cell monolayer model. The entire study process, including all three steps, could take approximately as little as six weeks to complete. With our extensive experience and innovative solutions, we can classify a broad range of drugs using in vitro methods alone. The classification includes experiments providing data on: GLP BCS classification of permeability and/or solubilityĪll experimental tests required will be performed under GLP.Quality permeability data will be obtained, including: This step includes the conduct of FDA-required experiments to establish the most cost-effective protocol for pivotal studies.
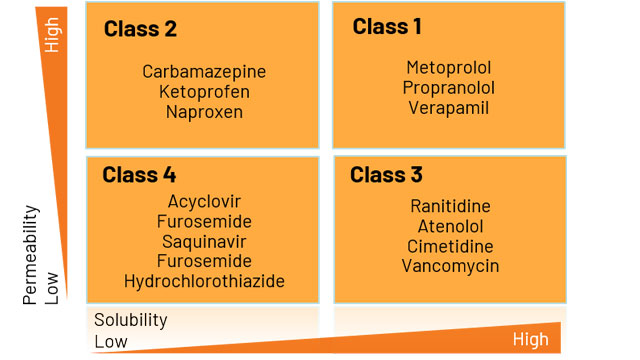
This includes a consultative support letter to regulatory authorities providing support to request the biowaiver approach. Is permeability greater than minoxidil?Īfter determining eligibility, we produce a report and expert statement.The main determinants in these experiments include: We can determine the biowaiver eligibility of your compound in 1-2 weeks using non-GMP material.Ī go/no go decision point – this step offers a prequalification and determination of the eligibility of the test compound for a BCS biowaiver. Use of this pathway can save significant time and money in drug development of both generics and new drugs and is endorsed by the World Health Organization to make generic drugs more readily available to the global population.Īt Absorption systems, we have over 20 years of experience working with BCS and have conducted hundreds of definitive BCS classification studies, resulting in numerous successful biowaivers. Clinical bioavailability or bioequivalence studies may not be necessary for regulatory submission of drugs within these classes. The FDA and the EMA grant biowaivers for both Class I and III compounds. The Biopharmaceutics Classification System (BCS) is a scientific framework that classifies drug substances based on their aqueous solubility and intestinal permeability.īased on the permeability and solubility, compounds are classified into four classes:įigure 1: Illustration of BSC classification classes based on solubility and permeability
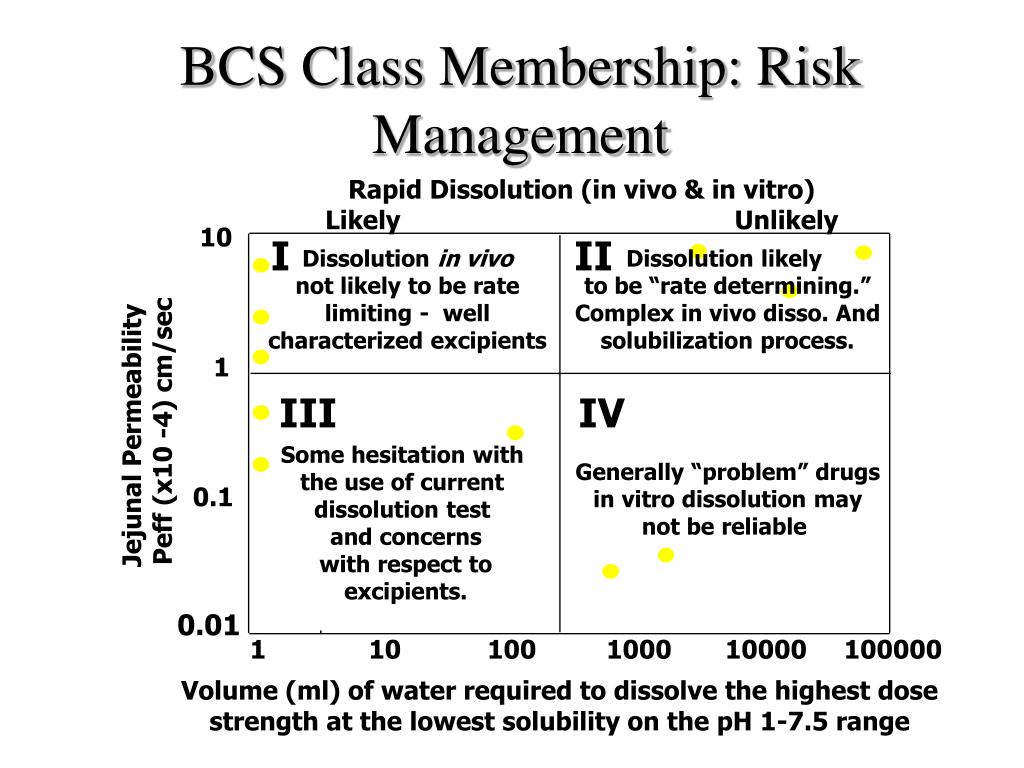
There are three main factors involved in the rate and extent of drug absorption in the body: the dissolution, aqueous solubility, and the permeability of the compound. To find out more about biowaivers, click here. To find out more about BCS, read the first blog here. This blog will cover what BCS classification studies entail, and how Absorption Systems can help you obtain all the right documentation to submit data on your drug to the regulatory agencies.
BCS CLASS 1 DRUGS SERIES
This series of blog posts aims to highlight BCS and BCS-based biowaiver applications as such an opportunity.

With most human trials on hold, drug developers and generics companies will be looking for new ways to progress product portfolios. The pharmaceutical industry is feeling the acute effects of disruption caused by the COVID-19 pandemic. Preclinical Toxicology Testing Services.Physicochemical Properties/Dose Selection.In Vitro Dissolution Absorption System (IDAS).Bioequivalence Testing (BCS, IVRT/IVPT).
